Big Brown Bat
Distribution, Abundance, and Seasonality
The big brown bat occurs throughout California. This widespread and abundant species has been recorded in virtually every North American vegetation type. Common to abundant in most of its range, the big brown bat is uncommon in hot desert habitats, and is absent only from the highest alpine meadows and talus slopes. Vagrant individuals may be seen in any habitat. During birth and development of young, sexes may be segregated. In southern Nevada (Burt 1934), males occur at higher elevations.
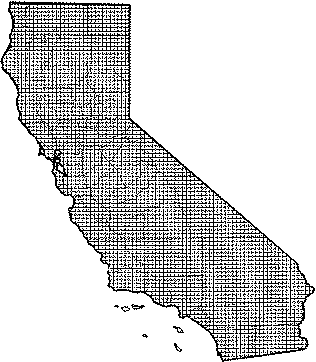
Range Map
Specific Habitat Requirements
Feeding: Feeds on a variety of flying insects. Large, hard-shelled prey, such as scarab beetles and other Coleoptera, usually are preferred. Diptera, Hymenoptera, and other insects are taken. Several studies report few moths eaten, in contrast to many other bat species. Feeds above open habitats among scattered trees and in residential areas, retracing its foraging route many times. Foraging height averages 6-9 m (20-30 ft) over open habitats. Some studies report foraging high over trees till dark, after which foraging height is reduced to low heights (to 1 m above ground). Foraging over water usually higher and further from shoreline than smaller myotis (Kurta 1982). Foraging flight is slow, straight, and steady. May carry food to the roost. A few terrestrial prey have been recorded, suggesting occasional feeding from the ground.
Cover: Uses buildings and other human-made structures for roosting to such an extent that natural roosting habits are poorly known. A few records include caves, mines, and trees. It has been suggested that it was formerly a tree-roosting species (Banfield 1974). McNab (1982) suggested that it roosts primarily in caves and buildings in the northern U.S., and in trees in the southern U.S. Small to moderate-sized groups roost together, usually less than 100 individuals. Night roosts are used, often in open buildings and structures. Winter roosts are largely unknown. Reported sites include cracks in caves, mines, and buildings. Hardy, occupying roosts with relatively low temperature and humidity. This species is intolerant of temperatures above 33Á C (92Á F).
Reproduction: Females and young form maternity colonies of 20-300 individuals. Such colonies are in warmer locations than ordinary roosting sites.
Water: Requires water. Consumes 0.64 ml/g/wk. This species has poor ability to concentrate urine, and probably needs to drink every other day.
Pattern: Prefers to forage over open areas, water sources, or among trees in fairly open stands.
Species Life History
Activity Patterns: Nocturnal. Hibernates. Time of emergence varies between studies, ranging from dusk to full darkness. Peak activity usually 1-2 hr after sunset. Activity of pregnant females is reduced. Stores up to 1/3 of weight in fat before hibernation. Though tolerant of cold, wakes if temperatures drop below 0ÁC (32ÁF), then shifts to warmer microclimates. Most authors report this species hibernates throughout its range (e.g., Barbour and Davis 1969), but Whitaker and Mumford (1972) reported that it was active throughout winter.
Seasonal Movements / Migration: Summer and winter ranges are the same, but there are local movements to suitable hibernacula. The longest such movement recorded was 290 km (180 mi) (Mills et al. 1975).
Home Range: There is little information on daily movements. Beer (1955) estimated home ranges of 111 km? (43 mi?). Capable of homing from 32 km (20 mi) on the same night as released, 400 km (250 mi) in 4-5 days. Does not home well at greater distances.
Territory: There is evidence of territoriality. Forages repeatedly over the same route. If a bat is removed, several days pass before the bat is replaced (Barbour and Davis 1969). A few aggressive encounters where foraging routes meet reported. Roosting sites colonial; may hibernate in tight clusters.
Reproduction: Most breeding is in fall, but ranges to March. This species has delayed fertilization. After a gestation period of 60 days, the young are born. In southern California (Krutzsch 1946), young born in late May-early June. The season of birth extends to July in other parts of the range. The single yearly litter ranges from 1-5. Generally, females west of the Great Plains give birth to a single young, while those from the Great Plains east give birth to 2 young (Barbour and Davis 1969). Lactating females have been found from May-August. The young reproduce in their first or second yr. First-year females have a lower rate of pregnancy than adults. Mortality in first yr is higher than that of adults, and is concentrated in the first winter. Mean annual survival of adults is usually high, but ranges from 10-77%. Maximum recorded longevity is 19 yr, but average age in a population usually is 2-3 yr.
Niche: Forages and roosts with other bat species. Relative to other bats, eats large, hard-shelled prey, and forages at moderate heights. Predators include owls, snakes, and hawks.
Comments: Although more tolerant of human disturbance than most bats, chronic disturbance may cause abandonment of a roost. Very sensitive to DDT and PCBs. Fat-soluble pesticides are sequestered in fat deposits in the fall and winter. In spring, when bats emerge from hibernation and fat reserves are depleted, bats may be exposed to fatal levels of pesticides (Lukens and Davis 1964). May carry rabies.
Sources & References
California Department of Fish and Game, 1999.
California's Wildlife, Sacramento, CA.
Written by: J. Harris, reviewed by: P. Brown, edited by: D. Alley, R. Duke
Banfield, A. W. F. 1974. The mammals of Canada. Univ. Toronto Press, Ontario. 438pp. Barbour, R. W., and W. H. Davis. 1969. Bats of America. Univ. of Kentucky Press, Lexington. 286pp. Beer, J. R. 1955. Survival and movements of banded big brown bats. J. Mammal. 36:242-248. Burt, W. H. 1934. The mammals of southern Nevada. Trans. San Diego Soc. Nat. Hist. 7:375-427. Christian, J. J. 1956. The natural history of a summer aggregation of the big brown bat, Eptesicus fuscus fuscus. Am. Midl. Nat. 55:66-95. Cockrum, E. L. 1955. Reproduction in North American bats. Trans. Kans. Acad. Sci. 58:487-511. Davis, W. H., R. W. Barbour, and M. D. Hassell. 1968. Colonial behavior of Eptesicus fuscus. J. Mammal. 49:51-62. Goehring, H. H. 1972. Twenty-year study of Eptesicus fuscus in Minnesota. J. Mammal. 53:201-207. Krutzsch, P. H. 1946. Some observations on the big brown bat in San Diego County, California. J. Mammal. 24:240-242. Kurta, A. 1982. Flight patterns of Eptesicus fuscus and Myotis lucifugus over a stream. J. Mammal. 63:335-337. Lukens, M. M., and W. H. Davis. 1964. Bats: sensitivity to DDT. Science 146:948. McNab, B. K. 1982. Physilological ecology. Pages 151-200 in T. H. Kunz, ed. Ecology of bats. Plenum Publ. Co., New York. 425pp. Mills, R. S., G. W. Barrett, and M. P. Farrell. 1975. Population dynamics of the big brown bat (Eptesicus fuscus) in southwestern Ohio. J. Mammal. 56:591-604. Whitaker, J. O., Jr., and R. E. Mumford. 1972. Notes on occurrence and reproduction of bats in Indiana. Proc. Indiana Acad. Sci. 81:376-383.
California Animal Facts | California's Wildlife